Modeling Movement Through Heterologous Biofilm Structures
• Provide insight into the biology of biofilms and understanding of the microbiome.• Lay the groundwork for studies ranging from translational applications such as nanoparticles for drug delivery, to basic research on how heterologous resident microbiota can reduce drug accessibility to intestinal tumors.
ABOUT
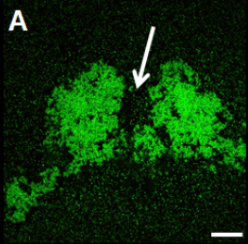
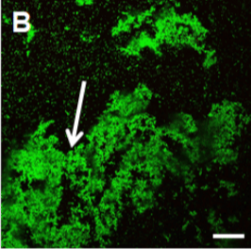
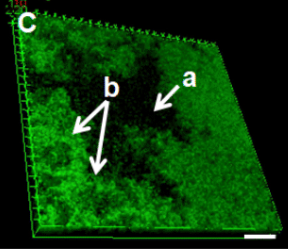
Biofilms, multicellular communities of bacteria encased in an extracellular matrix, are an essential part of most bacterial communities. The extracellular matrix can be composed of multiple materials such as extracellular DNA, proteins, polysaccharides, and amyloids. In humans, these communities range from pathogenic bacteria to commensal members of the microbiota. The treatment of pathogenic biofilms is difficult as associated bacteria are resistant to antibiotics > 1000-fold than non-biofilm associated planktonic counterparts. This leads to long-term, high-dose antibiotic treatment, which accelerates the emergence of multidrug-resistant bacteria, and in some cases, still ends in treatment failure.
SUMMARY
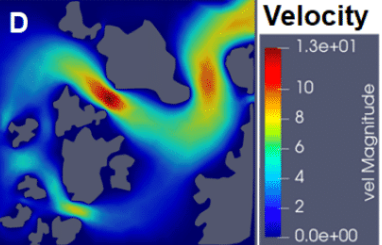
We use antibiotics and nanoparticles moving through biofilms as well as mixed species biofilms because this has implications for two important processes; the treatment of pathogenic biofilms and to determine if heterologous structures in microbiota help prevent the complete eradication of microbiota during antibiotic treatment. We use prototypical examples, Gram-positive and Gram-negative bacteria, as well as mixed viscous species and heterologous rigid structure species biofilms as microbiota models. Selected model systems, for example, mutants in matrix components needed for rigid structure formation, are used to experimentally inform the computational models regarding the contribution to antibiotic resistance.